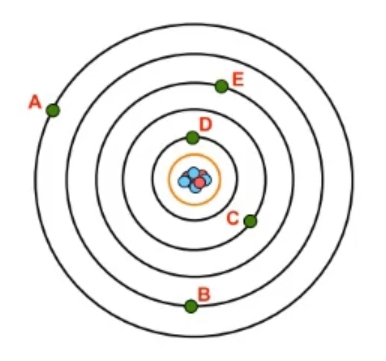
In atomic structure, the concept of shells, represented by the variable n, refers to the grouping of electrons that surround the nucleus of an atom. These shells are crucial as they relate to the potential energy of the electrons, indicating their energy of position. As the value of n increases, both the size and energy level of the atomic orbitals also increase. This relationship is directly tied to the periods or rows of the periodic table, where each period corresponds to a specific shell. For instance, the first row of the periodic table aligns with shell 1, the second row with shell 2, and so forth, up to the seventh row, which corresponds to shell 7.
Currently, the periodic table consists of seven rows, but it is important to recognize that it is a dynamic entity. Many elements in the seventh row have been discovered and named in recent years, highlighting the ongoing exploration and advancement in technology that may lead to the discovery of new elements. The potential for an eighth row, and beyond, exists as we continue to innovate and explore. The number of elements is theoretically limitless, constrained only by our imagination and ingenuity.
It is essential to note that the principal quantum number n must be a whole number, starting from 1 and extending to infinity. This means that while we currently recognize seven shells, the possibility of creating or discovering elements with more than seven shells exists. Thus, the relationship between the rows of the periodic table and the shell numbers of an atom is fundamental, with the understanding that the potential for new discoveries is boundless.