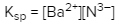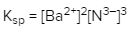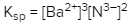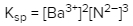Solubility is a fundamental chemical property that describes how well a solute can dissolve in a solvent. A key concept related to solubility is the solubility product constant, denoted as Ksp. This constant serves as the equilibrium constant specifically for the solubility of ionic solids. When examining the solubility of these solids, it is important to understand that solubility can also be expressed in terms of concentration or molarity, represented by the symbol M.
The relationship between Ksp and solubility is straightforward: a higher Ksp value indicates greater solubility of the ionic solid, meaning more of it can dissolve in the solvent. Conversely, a lower Ksp value signifies that the ionic solid is less soluble, resulting in a smaller amount dissolving in the solvent. This understanding of Ksp is crucial when analyzing the solubility behavior of ionic compounds in various solutions.