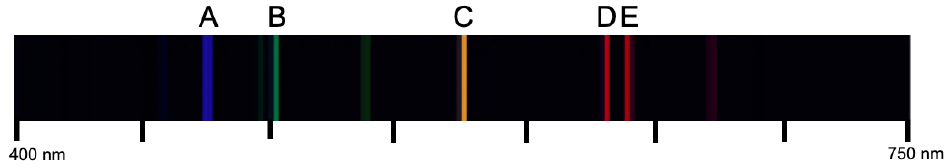
Emission spectra are created when emitted light from atoms is focused through a slit and then passed through a prism. In an atom, electrons occupy various energy levels or shells, theoretically infinite in number. When an electron transitions from a higher energy state to a lower one, it releases energy in the form of light. This process is crucial for understanding how light interacts with matter.
As the electron drops from a higher shell to a lower shell, it emits energy that can be captured and analyzed. The slit serves to narrow the emitted light, allowing closely packed wavelengths to be spread out. When this light passes through a prism, it is separated into its constituent wavelengths, resulting in an emission spectrum characterized by distinct lines. Each line corresponds to a specific wavelength of light emitted during the electron's transition.
The emission spectrum can be measured to determine the wavelengths of these lines, which can further be used to calculate the energy and frequency of the emitted light. The relationship between energy (E), frequency (f), and wavelength (λ) is given by the equation:
\[E = hf = \frac{hc}{\lambda}\]
where h is Planck's constant, approximately \(6.626 \times 10^{-34} \, \text{Js}\), and c is the speed of light, approximately \(3.00 \times 10^8 \, \text{m/s}\). Understanding emission spectra is essential for identifying elements and their properties, as each element emits light at characteristic wavelengths, forming a unique spectral fingerprint.