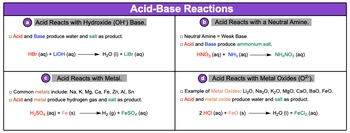
Acid-base reactions are fundamental chemical processes that involve the interaction of acids with various substances, leading to the formation of different products. One common type of reaction occurs when acids react with hydroxide bases, represented by the hydroxide ion (OH-). In this scenario, the acid donates a proton (H+), which combines with the hydroxide ion to produce water (H2O) and a salt, an ionic compound. For example, when hydrobromic acid (HBr) reacts with lithium hydroxide (LiOH), the products are water and lithium bromide (LiBr), an aqueous salt.
Another type of acid-base reaction involves neutral amines, which are weak bases containing nitrogen and hydrogen, or carbon, nitrogen, and hydrogen. A classic example is the reaction between nitric acid (HNO3) and ammonia (NH3). This reaction produces ammonium nitrate (NH4NO3), a salt formed from the combination of the acid and the amine.
Acids can also react with metals, such as sodium, potassium, magnesium, and zinc. In these reactions, the acid reacts with the metal to produce hydrogen gas (H2) and a salt. For instance, when sulfuric acid (H2SO4) reacts with iron (Fe), hydrogen gas is released, and iron(II) sulfate (FeSO4) is formed as a salt.
Lastly, acids can react with metal oxides, which include compounds like lithium oxide (Li2O) and iron(II) oxide (FeO). The reaction between an acid and a metal oxide results in the formation of water and a salt. For example, when hydrochloric acid (HCl) reacts with iron(II) oxide, it produces water and iron(II) chloride (FeCl2).
Understanding these patterns of acid-base reactions is crucial for predicting the products formed based on the type of acid and the reacting substance. Recognizing the nature of the reactants allows for effective identification of the resulting compounds in these chemical processes.