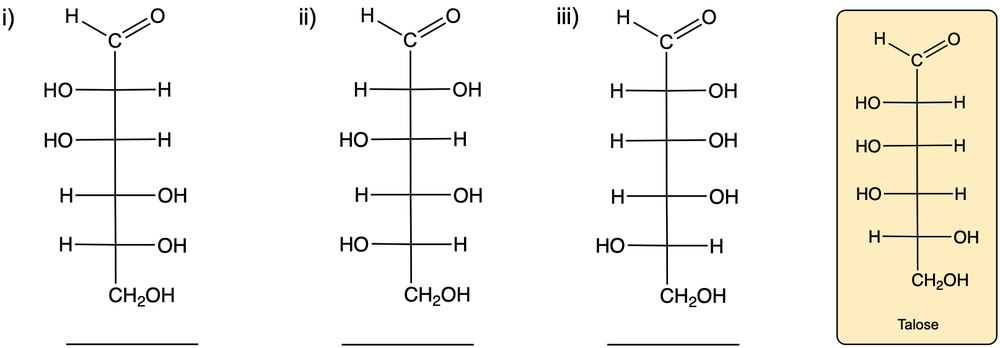
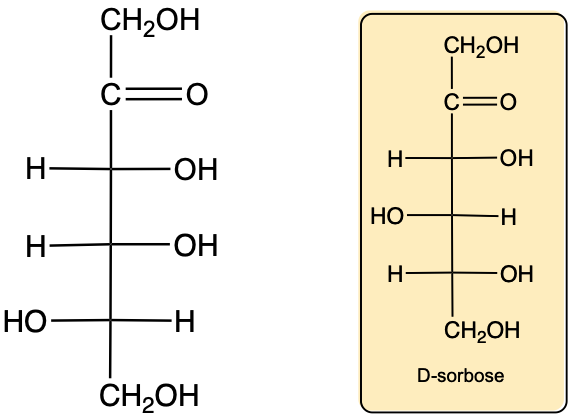
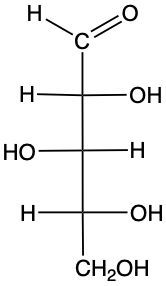
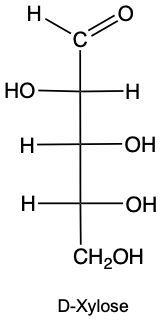
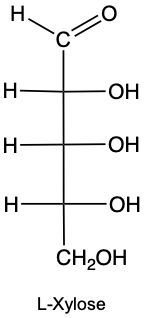
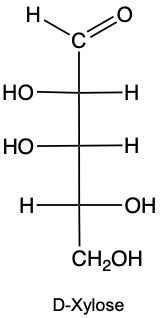
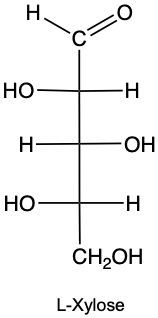
Monosaccharides, the simplest form of carbohydrates, can exist as either D or L enantiomers, which are distinguished by the configuration of the penultimate carbon, the last chiral carbon in the molecule. To determine whether a monosaccharide is a D or L sugar, one must examine the orientation of the hydroxyl group (–OH) attached to this carbon.
For a D enantiomer, if the hydroxyl group on the penultimate carbon is positioned on the right side, the sugar is classified as a D sugar. Conversely, if the hydroxyl group is on the left side, the sugar is identified as an L sugar. This distinction is crucial in carbohydrate chemistry, as the majority of naturally occurring carbohydrates are D sugars.
In summary, the classification of monosaccharides into D and L forms hinges on the orientation of the hydroxyl group at the last chiral center. Understanding this stereochemical configuration is essential for studying the properties and behaviors of carbohydrates in biological systems.