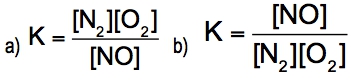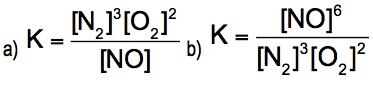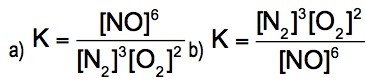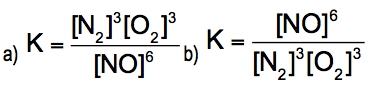The equilibrium constant, denoted as \( K \), is a crucial concept in chemical equilibrium, representing the ratio of the concentrations of products to reactants at a specific temperature. This relationship can be expressed mathematically as:
\[ K = \frac{[\text{Products}]}{[\text{Reactants}]} \]
Temperature plays a significant role in determining the value of \( K \); an increase in temperature generally leads to an increase in \( K \), while a decrease in temperature results in a lower \( K \). The magnitude of \( K \) provides insight into the position of equilibrium for a given reaction. When \( K > 1 \), it indicates that products are favored, suggesting that the reaction proceeds predominantly in the forward direction. For example, if the concentration of products is 10 and that of reactants is 1, then:
\[ K = \frac{10}{1} = 10 \]
This value, being greater than 1, confirms that the formation of products is favored. Conversely, when \( K < 1 \), reactants are favored, indicating that the reaction is more likely to proceed in the reverse direction. For instance, if the products are 1 and the reactants are 10, then:
\[ K = \frac{1}{10} = 0.1 \]
In this case, the reaction favors the reactants. When \( K = 1 \), it signifies that the concentrations of products and reactants are equal, indicating a state of balance in the reaction. For example, if both products and reactants are present in equal amounts, such as 10 each, then:
\[ K = \frac{10}{10} = 1 \]
It is important to note that the equilibrium constant \( K \) considers all states of matter except for solids and liquids, which are not included in the expression due to their constant concentrations in a given reaction. Understanding these principles is essential for predicting the behavior of chemical reactions under varying conditions.