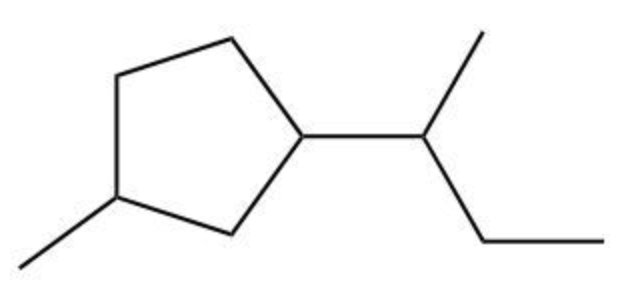
Cycloalkanes are a class of hydrocarbons characterized by their ring structure, which distinguishes them from linear alkanes. The naming of cycloalkanes follows a systematic approach similar to that of alkanes with substituents. The primary step in naming these compounds is to identify the parent chain, which is designated as a "cyclo" followed by the name of the alkane corresponding to the number of carbon atoms in the ring.
When naming cycloalkanes, it is essential to consider the location of any substituents attached to the ring. The substituents are numbered based on their position on the cycloalkane, starting from one of the carbon atoms in the ring. This numbering helps in providing a clear and unambiguous name for the compound. For example, in a cyclohexane with a methyl group, the compound would be named as methylcyclohexane, with the position of the methyl group indicated if necessary.
Understanding these naming conventions is crucial for accurately identifying and communicating the structure of cycloalkanes in organic chemistry. As you progress, you will encounter various types of cycloalkane compounds, each requiring careful application of these naming rules.