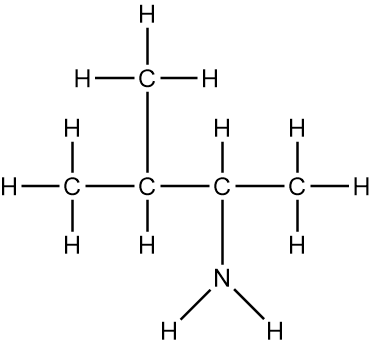
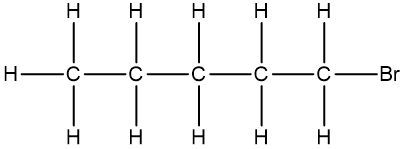
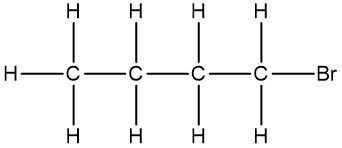
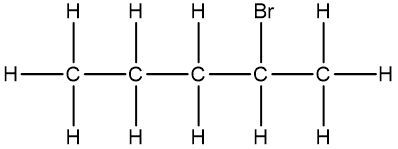
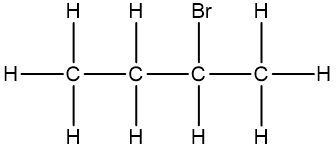
In chemistry, the condensed formula provides a simplified representation of a compound, illustrating how atoms are grouped without detailing every bond. For instance, in the condensed formula CH3, the three hydrogen atoms are grouped with the carbon atom, indicating that they are bonded together. Similarly, CH2 shows two hydrogen atoms bonded to a carbon atom, while CH represents a single carbon atom bonded to one hydrogen atom.
In contrast, the structural formula explicitly displays all the bonds between atoms, including carbon-carbon and carbon-hydrogen connections. This clarity is essential for understanding the molecular structure. For example, when transitioning from a structural formula to a condensed formula, we can group the atoms together. The structural representation of a molecule might show the bonds between carbons, but in the condensed version, we can write it as CH3CH2CH3, effectively condensing the information.
To further simplify, we can eliminate the visual representation of the carbon-carbon bonds, resulting in a fully condensed formula that still conveys the same information about the molecular structure. This method of representation is particularly useful for larger organic compounds, allowing chemists to quickly understand the composition and connectivity of the atoms involved.