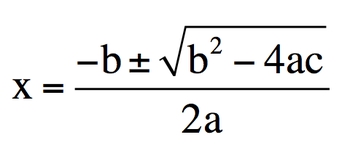The quadratic formula is a powerful tool used to solve algebraic equations of the form \( ax^2 + bx + c = 0 \), where \( a \), \( b \), and \( c \) are numerical coefficients, and \( x \) represents the variable we want to solve for. The formula is expressed as:
\( x = \frac{-b \pm \sqrt{b^2 - 4ac}}{2a} \)
This formula is particularly useful in various applications, including chemical equilibrium problems, where it helps determine equilibrium concentrations using ICE (Initial, Change, Equilibrium) charts. In these scenarios, the quadratic formula can yield two potential solutions for \( x \) due to the plus or minus sign. However, typically only one of these solutions is relevant, often the positive value, depending on the context of the problem.
To illustrate the application of the quadratic formula, consider the equation derived from a specific problem:
\( 4x^2 + 2.13 \times 10^{-4}x - 1.75 \times 10^{-5} = 0 \)
In this equation, \( a = 4 \), \( b = 2.13 \times 10^{-4} \), and \( c = -1.75 \times 10^{-5} \). Plugging these values into the quadratic formula, we first calculate the discriminant \( b^2 - 4ac \) to ensure it is non-negative, which is necessary for real solutions. The calculation proceeds as follows:
\( b^2 - 4ac = (2.13 \times 10^{-4})^2 - 4(4)(-1.75 \times 10^{-5}) \)
After determining the value of the discriminant, we can find the square root and substitute back into the quadratic formula. This will yield two potential solutions for \( x \): one from the addition and one from the subtraction of the square root term. For example, if the calculations yield:
\( x_1 = \frac{-2.13 \times 10^{-4} + 0.016735}{8} \approx 0.002065 \)
\( x_2 = \frac{-2.13 \times 10^{-4} - 0.016735}{8} \approx -0.002119 \)
In this case, \( x_1 \) is the viable solution, while \( x_2 \) may be discarded based on the context of the problem, particularly in chemical equilibrium scenarios where negative concentrations are not physically meaningful.
Understanding the quadratic formula and its application in solving equations is crucial, especially in fields like chemistry, where it aids in analyzing equilibrium states and concentrations effectively.