1. Matter and Measurements
Density of Non-Geometric Objects
1. Matter and Measurements
Density of Non-Geometric Objects - Video Tutorials & Practice Problems
On a tight schedule?
Get a 10 bullets summary of the topicThe Density of non-geometric objects can be determined using water displacement.
Calculating Density
1
concept
Density of Non-Geometric Objects Concept and Example
Video duration:
3mPlay a video:
2
Problem
ProblemA piece of unknown solid weighs approximately 0.045 lbs. When a scientist places it in a glass beaker the water level increases from 200 mL to 260 mL. What is the density of the unknown solid in g/mL?
A
0.5 g/mL
B
0..2 g/mL
C
0.0007 g/mL
D
2.9 g/mL
E
0.3 g/mL
3
Problem
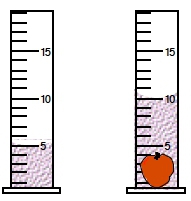
ProblemIf an irregularly shaped apple possesses a density of 0.96 g/cm3, what is its mass in milligrams? (The volume of the given cylinders are in mL).
A
4000 mg
B
5000 mg
C
0.0048 mg
D
4800 mg
E
5300 mg
Do you want more practice?
We have more practice problems on Density of Non-Geometric Objects
Your GOB Chemistry tutor
Additional resources for Density of Non-Geometric Objects
PRACTICE PROBLEMS AND ACTIVITIES (5)
- Determine the density (g/mL) for each of the following: c. A gem has a mass of 4.50 g. When the gem is placed...
- Use the density values in TABLE 2.8 to solve each of the following problems: a. A graduated cylinder contains...
- The water level in a graduated cylinder initially at 215 mL rises to 285 mL after a piece of lead is submerged...
- A graduated cylinder contains 155 mL of water. A 15.0-g piece of iron and a 20.0-g piece of lead are added. Wh...
- A 50.0-g silver object and a 50.0-g gold object are both added to 75.5 mL of water contained in a graduated cy...