What energy requirements must be met in order for a reaction to be favorable?

The electron-transport chain uses several different metal ions, especially iron, copper, zinc, and manganese. Why are metals used frequently in these two pathways? What can metals do better than organic biomolecules?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
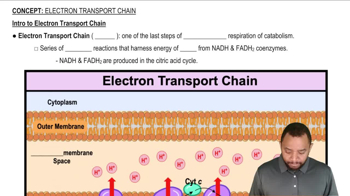
Electron Transport Chain (ETC)
Role of Metal Ions in Biochemical Reactions

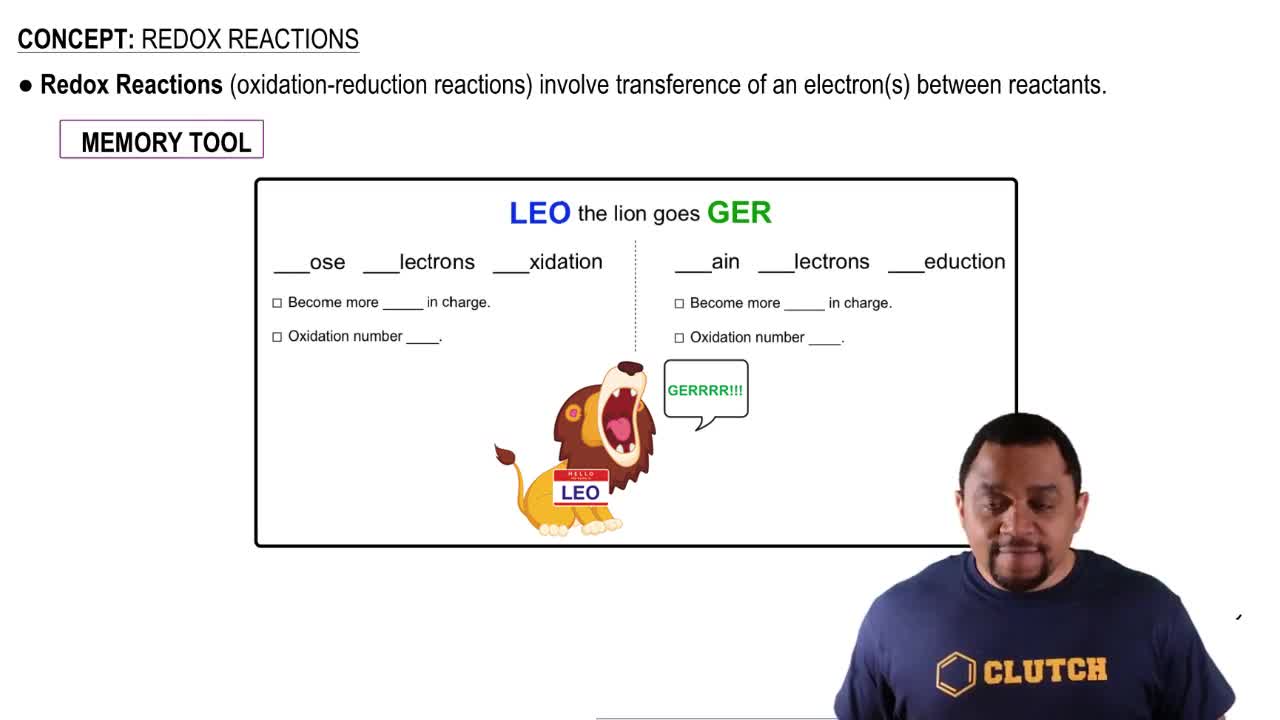
Redox Reactions
The reaction that follows is catalyzed by isocitrate dehydrogenase and occurs in two steps, the first of which (step A) is formation of an unstable intermediates (shown in brackets).
d. To what class of enzymes does isocitrate dehydrogenase, the enzyme that catalyzes this reaction, belong?
The following reactions occur during the catabolism of acetyl-CoA. Which are exergonic? Which is endergonic? Which reaction produces a phosphate that later yields energy by giving up a phosphate group?
c. L-Malate + NAD+ → Oxaloacetate + NADH + H+
∆G = +17 kcal/mol (+129.3 kJ/mol)
Why is ∆G a useful quantity for predicting the favorability of biochemical reactions?
The reaction that follows is catalyzed by isocitrate dehydrogenase and occurs in two steps, the first of which (step A) is formation of an unstable intermediates (shown in brackets).
b. In which step is CO2 evolved and a hydrogen ion added?
The reaction that follows is catalyzed by isocitrate dehydrogenase and occurs in two steps, the first of which (step A) is formation of an unstable intermediates (shown in brackets).
c. Which of the structures shown can be described as a β-keto acid?
