The carbonyl group can be reduced by addition of a hydride ion (H–) and (H+) a proton. Removal of H– and H+ from an alcohol results in a carbonyl group.
a. To which atom of the carbonyl is the hydride ion added and why?

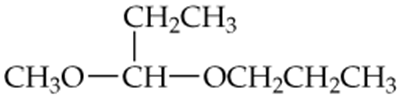
 Verified step by step guidance
Verified step by step guidance

The carbonyl group can be reduced by addition of a hydride ion (H–) and (H+) a proton. Removal of H– and H+ from an alcohol results in a carbonyl group.
a. To which atom of the carbonyl is the hydride ion added and why?
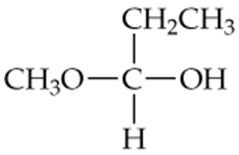
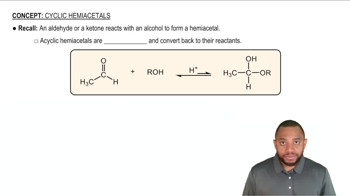
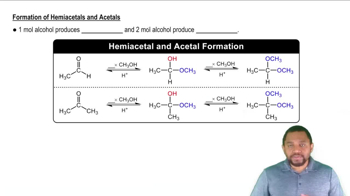
Draw the structures of the hemiacetals or hemiketals formed in these reactions:
b.
The carbonyl group can be reduced by addition of a hydride ion (H–) and (H+) a proton. Removal of H– and H+ from an alcohol results in a carbonyl group.
b. In the reaction, indicate which direction represents reduction and which represents oxidation.
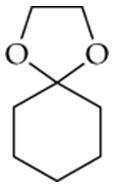
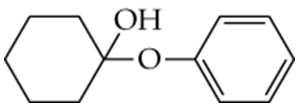
Determine whether the following compounds are acetals or ketals. Draw the structure of the aldehyde or ketone it came from.
c.
A fundamental difference between aldehydes and ketones is that one can be oxidized to carboxylic acids but the other cannot. Which is which? Give an example of a test to differentiate aldehydes from ketones.