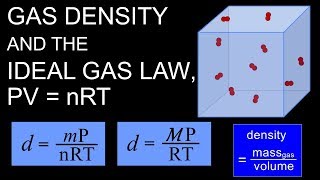
7. Gases
The Ideal Gas Law Derivations
Problem 48b
Textbook Question
Textbook QuestionAssume that you have a cylinder with a movable piston. What would happen to the gas pressure inside the cylinder if you were to do the following? (c) Decrease the volume by 45% at constant T
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
634
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos