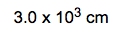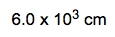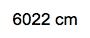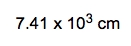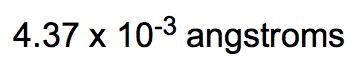To determine the mass of a cube of silver measuring 0.56 meters on each side, we start by recognizing the density of silver, which is 10.5 grams per cubic centimeter. The first step is to convert the dimensions of the cube from meters to centimeters, as the density is given in grams per cubic centimeter. Since 1 meter equals 100 centimeters, the side length of the cube in centimeters is:
Next, we calculate the volume of the cube using the formula for volume:
Now that we have the volume, we can use the density to find the mass. The formula relating mass, density, and volume is:
Substituting the known values:
To convert grams to kilograms, we use the conversion factor where 1 kilogram equals 1000 grams:
Considering significant figures, the original measurement of 0.56 meters has 2 significant figures, while the density of silver has 3 significant figures. Therefore, we round our final answer to 2 significant figures, resulting in:
This example illustrates the importance of dimensional analysis and unit conversion in solving problems involving density and volume. By carefully managing units and significant figures, we can arrive at a precise and accurate answer.