In the study of alcohol reactions, particularly oxidation reactions, it is essential to understand the role of oxidizing agents such as Sodium Dichromate or Potassium Dichromate. These agents are interchangeable due to both sodium and potassium being Group 1A elements, but the key component responsible for oxidation is the dichromate ion. When these oxidizing agents are dissolved in sulfuric acid, they react with alcohols to facilitate oxidation.
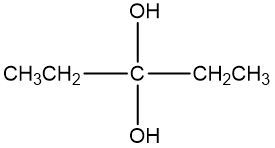
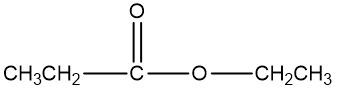
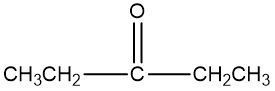
An oxidizing agent is defined as a compound that promotes the oxidation of alcohols by increasing the number of carbon-oxygen bonds while ensuring that carbon-carbon bonds remain intact. This process is crucial in organic chemistry, particularly when transforming alcohols into various functional groups. For instance, an alcohol can be oxidized to form a carbonyl compound, which may manifest as either an aldehyde or a ketone. The oxidation can progress further, leading to the formation of a carboxylic acid.
To summarize, the oxidation pathway typically follows this sequence: alcohols are converted to aldehydes or ketones, which can then be further oxidized to yield carboxylic acids. It is important to adhere to the principle of maximizing carbon-oxygen bonds without disrupting carbon-carbon bonds to achieve the desired oxidized products effectively.