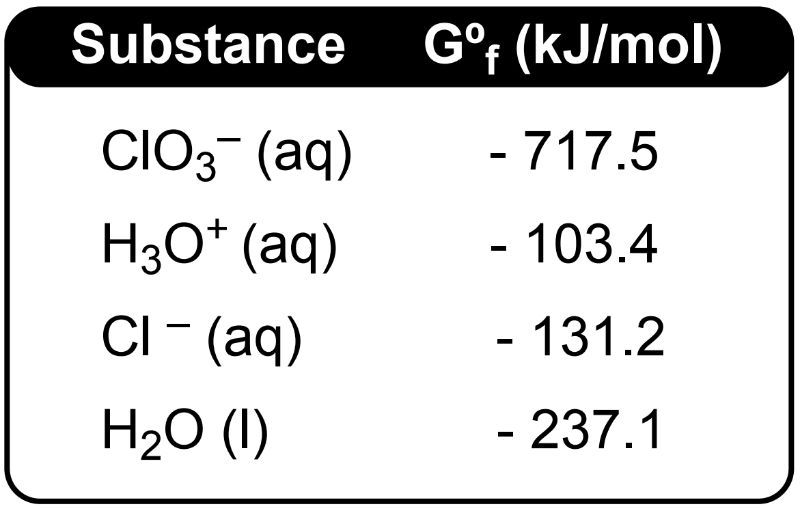
In the context of electrochemical cells, the concept of Gibbs free energy, denoted as ΔG°, signifies the maximum electrical work that can be performed by the system under standard conditions. The relationship between spontaneity, Gibbs free energy, and standard cell potential is encapsulated in the equation:
\[\Delta G° = -n \cdot F \cdot E°_{cell}\]
In this equation, ΔG° is expressed in kilojoules, while the variables represent the following:
- n: This is the number of moles of electrons transferred during the redox reaction. In such reactions, one species undergoes oxidation (loses electrons) and another undergoes reduction (gains electrons).
- F: Known as Faraday's constant, it is valued at 96,485 coulombs per mole of electrons. This constant is crucial for converting the amount of charge to the number of moles of electrons.
- E°cell: This represents the standard cell potential, measured in volts (V). It indicates the driving force behind the electrochemical reaction.
This equation effectively links the thermodynamic properties of the reaction (Gibbs free energy) with the electrochemical characteristics (cell potential), illustrating how the spontaneity of a reaction can be predicted based on these parameters. A negative ΔG° indicates a spontaneous reaction, while a positive ΔG° suggests non-spontaneity, thereby highlighting the importance of understanding these relationships in electrochemistry.