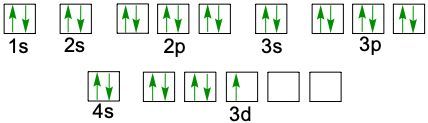
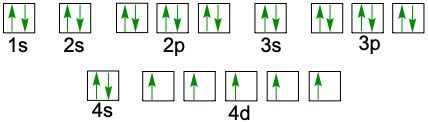
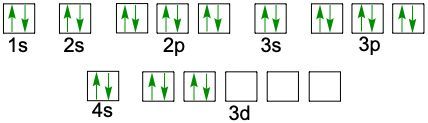
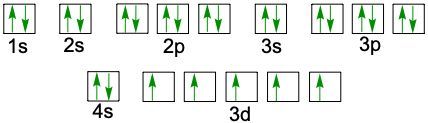
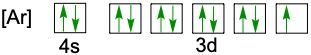
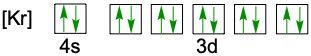
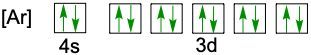
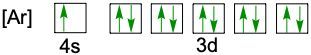
Understanding electron configuration is essential for grasping how electrons are distributed in an atom. The Aufbau principle guides this process, stating that electrons fill orbitals starting from the lowest energy level to the highest. This means we begin with the 1s orbital, followed by 2s, 2p, and so forth, ensuring that lower energy orbitals are filled before higher ones.
To visualize this filling order, the Aufbau diagram is a helpful tool. It illustrates the sequence in which orbitals are filled: starting from 1s, we progress to 2s, then 2p, 3s, 3p, 4s, and continue this pattern through 3d, 4p, 5s, and up to 8s. The diagram also includes the transition through 3d to 6d and 4f to 5f orbitals, providing a comprehensive view of the electron filling process.
Another effective method for determining electron configurations is by utilizing the periodic table. Each element's position on the table corresponds to its electron configuration, allowing for a more intuitive understanding of how electrons are arranged in various elements and ions. This approach not only simplifies the process but also reinforces the connection between an element's properties and its electron configuration.