23. Chemistry of the Nonmetals
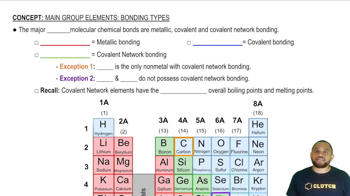
Main Group Elements: Bonding Types
Problem 96
Textbook Question
Textbook QuestionFrom the given molar mass and empirical formula of several compounds, find the molecular formula of each compound. a. C4H9, 114.22 g/mol
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
1297
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos