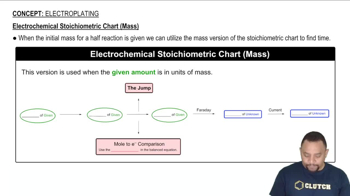
20. Electrochemistry
Electroplating
Problem 124
Textbook Question
Textbook QuestionA metal forms the fluoride MF3. Electrolysis of the molten fluo- ride by a current of 3.86 A for 16.2 minutes deposits 1.25 g of the metal. Calculate the molar mass of the metal.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
633
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos