2. Atoms & Elements
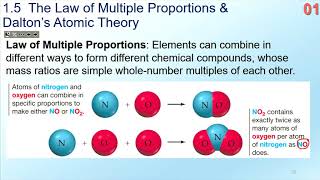
Law of Multiple Proportions
Problem 77
Textbook Question
Textbook QuestionWhat is the law of multiple proportions, and how does Dalton's atomic theory account for it?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
514
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos