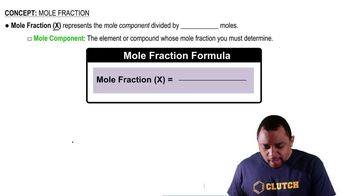
Textbook Question
At 63.5 °C, the vapor pressure of H2O is 175 torr, and that of ethanol (C2H5OH) is 400 torr. A solution is made by mixing equal masses of H2O and C2H5OH. (a) What is the mole fraction of ethanol in the solution?
2059
views

 Verified step by step guidance
Verified step by step guidance