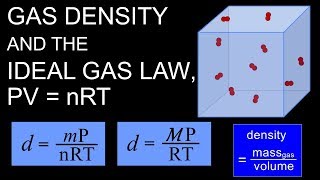
7. Gases
The Ideal Gas Law Derivations
Problem 49a
Textbook Question
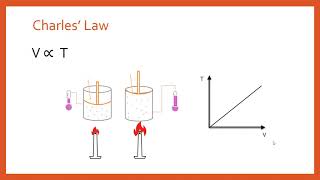
Textbook QuestionAssume that you have a cylinder with a movable piston. What would happen to the gas volume of the cylinder if you were to do the following? (a) Halve the Kelvin temperature while holding the pressure constant
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
426
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos