2. Atoms & Elements
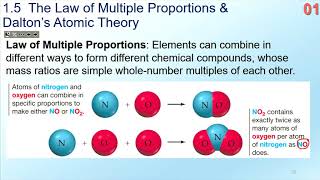
Law of Multiple Proportions
Problem 35
Textbook Question
Textbook QuestionTwo different compounds containing osmium and oxygen have the following masses of oxygen per gram of osmium: 0.168 and 0.3369 g. Show that these amounts are consistent with the law of multiple proportions.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1165
views
4
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos