17. Acid and Base Equilibrium
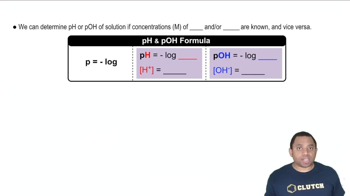
The pH Scale
Problem 55
Textbook Question
Textbook QuestionCalculate the pH of each acid solution. Explain how the resulting pH values demonstrate that the pH of an acid solution should carry as many digits to the right of the decimal place as the number of significant figures in the concentration of the solution. [H3O+] = 0.044 M [H3O+] = 0.045 M [H3O+] = 0.046 M
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1107
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos