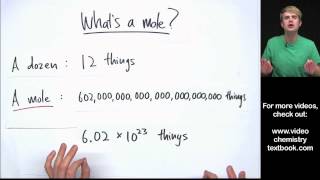2. Atoms & Elements
Mole Concept
Problem 38
Textbook Question
Textbook Question(a) What is the mass, in grams, of 1.223 mol of iron(III) sulfate? (b) How many moles of ammonium ions are in 6.955 g of ammonium carbonate? (c) What is the mass, in grams, of 1.50 * 1021 molecules of aspirin, C9H8O4?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
721
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos