2. Atoms & Elements
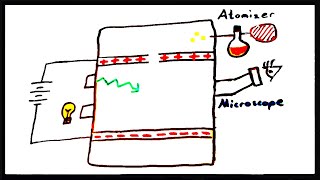
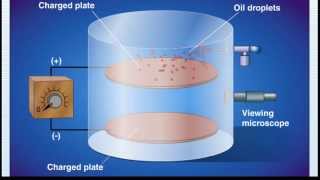
Millikan Oil Drop Experiment
Problem 90
Textbook Question
Textbook QuestionWhich of the following charges is not possible for the over-all charge on an oil droplet in Millikan's experiment? For this problem, we'll round the currently accepted charge of an electron to 1.602 * 10-19 C. (a) -1.010 * 10-18 C (b) -8.010 * 10-19 C (c) -2.403 * 10-18 C
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
775
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos