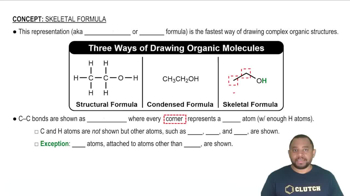
Textbook Question
Which type of electromagnetic radiation will cause the greatest number of electrons to be ejected from zinc metal with a work function of 350 kJ/mol? (LO 5.4, 5.5)(a) Dim light with a wavelength of 320 nm (b) Dim light with a wavelength of 360 nm (c) Bright light with a wavelength of 360 nm (d) Bright light with a wavelength of 375 nm
806
views
1
rank