7. Gases
Partial Pressure
Problem 62b
Textbook Question
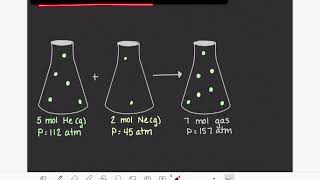
Textbook QuestionA gas mixture with a total pressure of 745 mmHg contains each of the following gases at the indicated partial pressures: CO2, 125 mmHg; Ar, 214 mmHg; and O2, 187 mmHg. The mixture also contains helium gas. What is the partial pressure of the helium gas? What mass of helium gas is present in a 12.0-L sample of this mixture at 273 K?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
5682
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos