3. Chemical Reactions
Molecular Formula
Problem 53a
Textbook Question
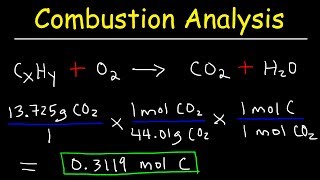
Textbook QuestionDetermine the empirical and molecular formulas of each of the following substances: (a) Styrene, a compound used to make Styrofoam® cups and insulation, contains 92.3% C and 7.7% H by mass and has a molar mass of 104 g/mol.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
1166
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos