6. Chemical Quantities & Aqueous Reactions
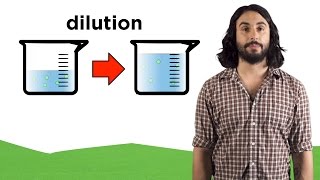
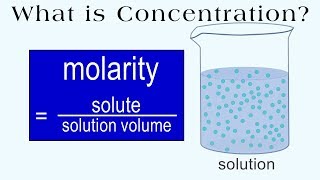
Molarity
Problem 49b
Textbook Question
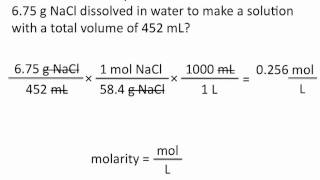
Textbook QuestionThe density of acetonitrile 1CH3CN2 is 0.786 g>mL and the density of methanol 1CH3OH2 is 0.791 g>mL. A solution is made by dissolving 22.5 mL of CH3OH in 98.7 mL of CH3CN. (c) Assuming that the volumes are additive, what is the molarity of CH3OH in the solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
999
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos