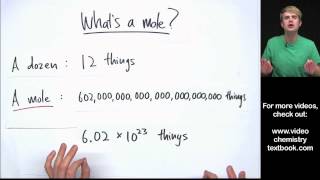2. Atoms & Elements
Mole Concept
Problem 122a
Textbook Question
Textbook QuestionThe U.S. Environmental Protection Agency (EPA) sets limits on healthful levels of air pollutants. The maximum level that the EPA considers safe for lead air pollution is 1.5 mg>m3. If your lungs were filled with air containing this level of lead, how many lead atoms would be in your lungs? (Assume a total lung volume of 5.50 L.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1301
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos