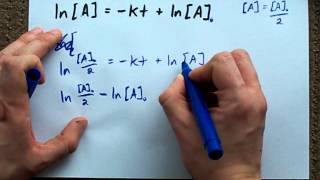21. Nuclear Chemistry
Rate of Radioactive Decay

Get help from an AI Tutor
Ask a question to get started.
Problem 60
Textbook Question
Textbook QuestionRadioactive decay exhibits a first-order rate law, rate = -∆N/∆t = kN, where N denotes the number of radio-active nuclei present at time t. The half-life of strontium-90, a dangerous nuclear fission product, is 29 years. (a) What fraction of the strontium-90 remains after three half-lives?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
280
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos