6. Chemical Quantities & Aqueous Reactions
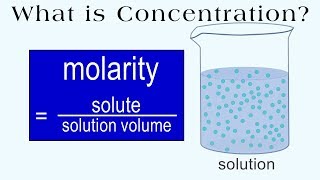
Molarity
Problem 77
Textbook Question
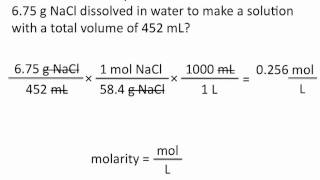
Textbook QuestionPure acetic acid, known as glacial acetic acid, is a liquid with a density of 1.049 g/mL at 25 C. Calculate the molarity of a solution of acetic acid made by dissolving 20.00 mL of glacial acetic acid at 25 C in enough water to make 250.0 mL of solution.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
4221
views
2
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos