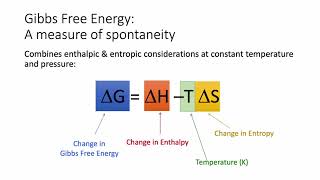
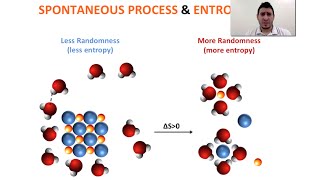
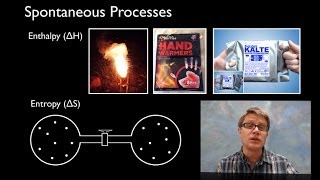
19. Chemical Thermodynamics
Spontaneous vs Nonspontaneous Reactions

Get help from an AI Tutor
Ask a question to get started.
Problem 126a
Textbook Question
Textbook QuestionTrouton's rule says that the ratio of the molar heat of vapor-ization of a liquid to its normal boiling point (in kelvin) is approximately the same for all liquids: ∆Hvap/Tbp ≈ 88 J/(K*mol) (c) Which of the liquids in the table deviate(s) from Trouton's rule? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
350
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos