1. Intro to General Chemistry
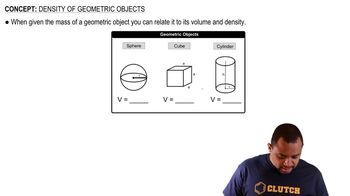
Density of Geometric Objects
Problem 130
Textbook Question
Textbook QuestionLead metal can be extracted from a mineral called galena, which contains 86.6% lead by mass. A particular ore contains 68.5% galena by mass. If the lead can be extracted with 92.5% efficiency, what mass of ore is required to make a lead sphere with a 5.00-cm radius?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1215
views
1
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos