
 McMurry 8th Edition
McMurry 8th Edition Ch.6 - Ionic Compounds: Periodic Trends and Bonding Theory
Ch.6 - Ionic Compounds: Periodic Trends and Bonding Theory Problem 12
Problem 12For a multielectron atom, a 3s orbital lies lower in energy than a 3p orbital because (LO 5.16)(a) a 3p orbital has more nodal surfaces than a 3s orbital. (b) an electron in a 3p orbital has a higher probability of being closer to the nucleus than an electron in a 3s orbital.(c) inner electrons shield electrons in a 3p orbital more effec-tively than electrons in a 3s orbital. (d) the energy of the electron can be spread between three 3p orbitals instead of only one 3s orbital.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Orbital Energy Levels
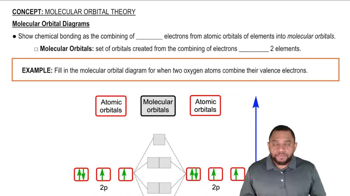
Shielding Effect
Nodal Surfaces

Predict the formula of the ionic compound that forms between potassium and sulfur. (LO 6.11) (a) KS (b) KS2 (c) K2S2 (d) K2S
Which molecular scale image best represents the ionic com-pound that forms between cesium and chlorine? (Cesium is represented by red circles, and chlorine is represented by blue circles.) (LO 6.12) (a)
(b)
(c)
(d)
Given the following information, construct a Born–Haber cycle to calculate the lattice energy of CaCl2(s). (LO 6.13)
Net energy change for the formation of CaCl2(s) form Ca(s) and Cl2(g) = -795.4 kJ/mol
Heat of sublimation for Ca(s) = +178 kJ/mol
Ei1 for Ca(s) = +590 kJ/mol
Ei2 for Ca(g) = +1145 kJ/mol
Bond dissociation energy for Cl2(g) = +243 kJ/mol
Eea1 for Cl(g) = -348.6 kJ/mol
(a) 2603 kJ/mol (b) 2254 kJ/mol (c) 2481 kJ/mo (d) 1663 kJ/mol