1. Intro to General Chemistry
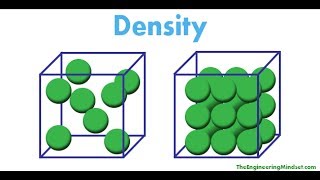
Density
Problem 93
Textbook Question
Textbook QuestionYou are assigned the task of separating a desired granular material with a density of 3.62 g>cm3 from an undesired granular material that has a density of 2.04 g>cm3. You want to do this by shaking the mixture in a liquid in which the heavier material will fall to the bottom and the lighter material will float. A solid will float on any liquid that is more dense. Using an Internet-based source or a handbook of chemistry, find the densities of the following substances: carbon tetrachloride, hexane, benzene, and diiodomethane. Which of these liquids will serve your purpose, assuming no chemical interaction takes place between the liquid and the solids?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
668
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos