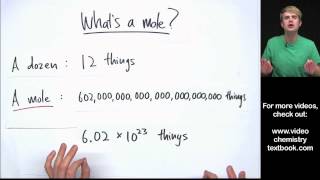2. Atoms & Elements
Mole Concept
Problem 66
Textbook Question
Textbook QuestionHow many molecules (or formula units) are in each sample? a. 85.26 g CCl4 b. 55.93 kg NaHCO3 c. 119.78 g C4H10 d. 4.59 * 105 g Na3PO4
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1106
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos