Textbook Question
Assign oxidation numbers to each element in the following ions. (f) V2O7 4-
312
views

 Verified step by step guidance
Verified step by step guidance
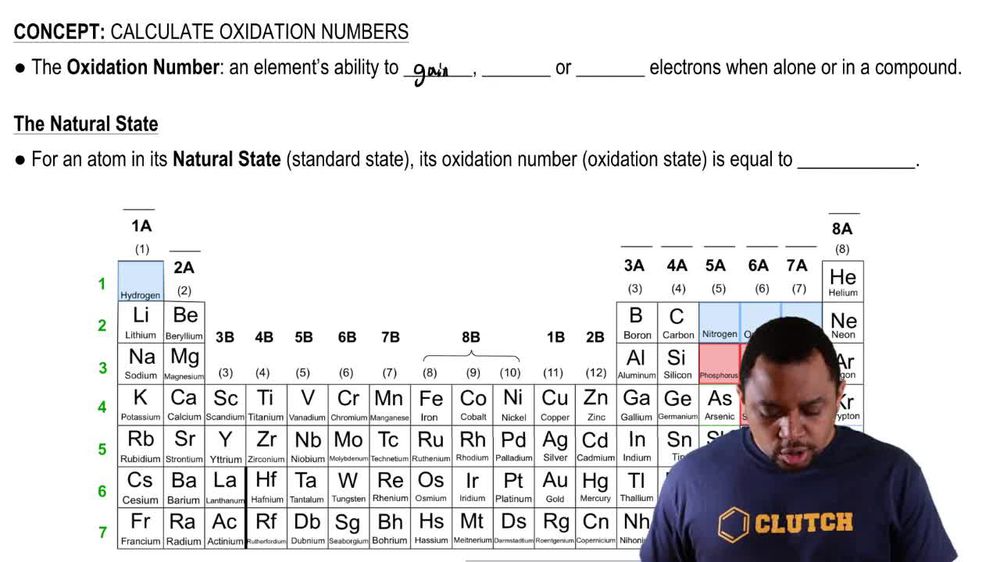

Assign oxidation numbers to each element in the following ions. (f) V2O7 4-
Nitrogen can have several different oxidation numbers ranging in value from -3 to +5. (a) Write the formula and give the name of the nitrogen oxide compound in which nitrogen has an oxidation number of +1, +2, +4, and +5.
Nitrogen can have several different oxidation numbers ranging in value from -3 to +5. (b) Based on oxidation numbers, which nitrogen oxide from part (a) cannot react with molecular oxygen?
Phosphorus can have several different oxidation numbers ranging in value from -3 to +5. (b) Based on oxidation numbers, which phosphorus oxide compound from part (a) was formed by combustion with a limited supply of oxygen?