1. Intro to General Chemistry
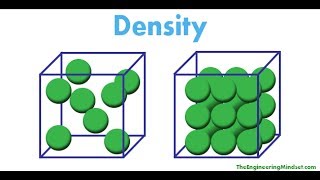
Density
Problem 90
Textbook Question
Textbook QuestionGold is alloyed (mixed) with other metals to increase its hardness in making jewelry. (b) The relative amount of gold in an alloy is commonly expressed in units of carats. Pure gold is 24 carat, and the percentage of gold in an alloy is given as a percentage of this value. For example, an alloy that is 50% gold is 12 carat. State the purity of the gold jewelry in carats.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
1230
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos