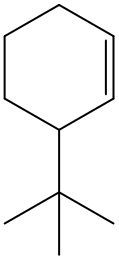
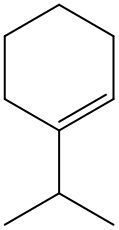
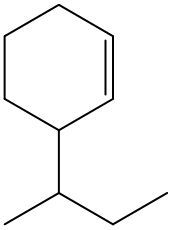
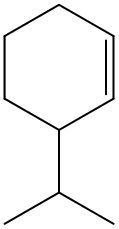
Alkenes are hydrocarbons characterized by the presence of a carbon-carbon double bond (C=C). When naming alkenes, the suffix changes from -ane (used for alkanes) to -ene to reflect this modification. This naming convention also requires specifying the location of the double bond and any substituents attached to the carbon chain.
Geometric isomerism is a key concept in understanding alkenes, as it arises from the restricted rotation around the double bond due to the presence of a pi bond. This restriction leads to different spatial arrangements of substituents, which can be classified as either cis or trans. The terms cis and trans are used when there are two different groups attached to the double-bonded carbons. If the two groups are on the same side of the double bond, the configuration is termed cis. Conversely, if the groups are on opposite sides, it is referred to as trans.
For example, consider two double-bonded carbon atoms with two methyl groups attached. If both methyl groups are positioned on the same side of the double bond, the compound is classified as a cis isomer. If one methyl group is above and the other is below the double bond, the compound is classified as a trans isomer. It is important to note that in cyclic alkenes, the cis/trans nomenclature does not apply, as the ring structure does not allow for the same type of spatial orientation.
In summary, when naming alkenes, remember to modify the suffix to -ene, indicate the position of the double bond, and use the terms cis or trans to describe the arrangement of substituents around the double bond, provided there are two different groups present.