The concept of electron geometry is essential in understanding molecular shapes, focusing on the arrangement of electron groups around a central atom. Electron groups include both lone pairs and bonding pairs, which are treated similarly in this context. The number of electron groups can be 2, 3, or 4, each leading to distinct geometrical arrangements.
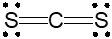
When there are 2 electron groups, the geometry is classified as linear. This can be visualized as two points connected in a straight line, emphasizing the simplicity of the arrangement. The linear geometry is characterized by a bond angle of 180 degrees.
For 3 electron groups, the geometry is termed trigonal planar. The prefix "tri-" indicates three groups, which can be arranged in a flat plane. This configuration typically results in bond angles of 120 degrees. It is important to note that with 3 electron groups, there are variations: you can have three surrounding atoms or two surrounding atoms with one lone pair.
When the central atom has 4 electron groups, the geometry becomes tetrahedral. The prefix "tetra-" signifies four groups, leading to a three-dimensional shape with bond angles of approximately 109.5 degrees. Similar to the trigonal planar arrangement, tetrahedral geometry can also accommodate lone pairs, resulting in variations such as two surrounding atoms and two lone pairs.
In summary, the electron geometries corresponding to 2, 3, and 4 electron groups are linear, trigonal planar, and tetrahedral, respectively. Understanding these geometries is crucial for predicting molecular shapes and their corresponding properties.