The ideal gas law, represented by the equation \( PV = nRT \), describes the relationship between pressure (P), volume (V), number of moles (n), the gas constant (R), and temperature (T). Understanding how these variables interact is crucial for solving problems related to gases.
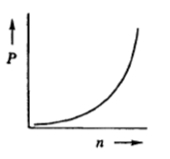
When analyzing the relationships between these variables, we can identify both direct and inverse proportionalities. For instance, if we focus on pressure and volume, we can rearrange the ideal gas law to express pressure as \( P = \frac{nRT}{V} \). This indicates that pressure is inversely proportional to volume; as volume increases, pressure decreases, and vice versa. This inverse relationship is significant because it highlights how changes in one variable affect the other when the number of moles and temperature remain constant.
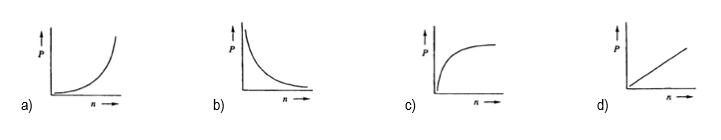
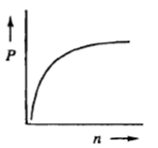
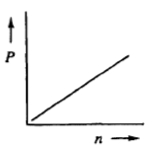
Next, we can explore the relationship between pressure and the number of moles. By isolating pressure, we find that \( P \) is directly proportional to \( n \) when volume and temperature are held constant. This means that increasing the number of moles will lead to an increase in pressure, demonstrating a direct relationship.
Similarly, when examining pressure and temperature, we find that they are also directly proportional. If we increase the temperature while keeping volume and moles constant, the pressure will increase as well.
Now, let’s consider volume in relation to the number of moles. Here, volume is directly proportional to moles; increasing the volume will result in an increase in the number of moles, assuming pressure and temperature are constant.
Finally, the relationship between volume and temperature also reveals a direct proportionality. As volume increases, temperature increases, provided that pressure and moles remain unchanged.
In summary, the ideal gas law illustrates that pressure and volume have an inverse relationship, while pressure and moles, pressure and temperature, volume and moles, and volume and temperature all exhibit direct relationships. Understanding these interactions is essential for predicting how changes in one variable will affect others in gas-related scenarios.