19. Chemical Thermodynamics
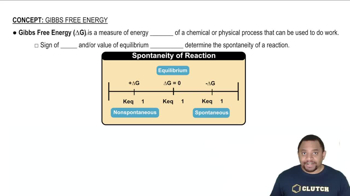
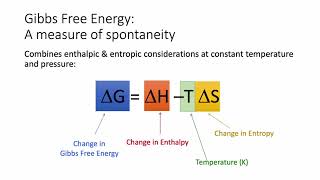
Gibbs Free Energy
Problem 89
Textbook Question
Textbook QuestionConsider this reaction occurring at 298 K: N2O( g) + NO2( g) ∆ 3 NO( g) a. Show that the reaction is not spontaneous under standard conditions by calculating ΔGrxn ° .
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
1303
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos