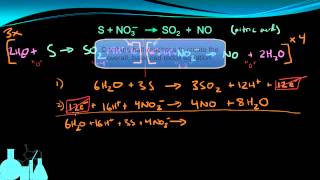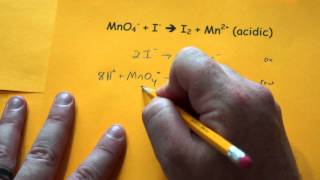6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Acidic Solutions
Problem 107
Textbook Question
Textbook QuestionThe discovery of hafnium, element number 72, provided a controversial episode in chemistry. G. Urbain, a French chemist, claimed in 1911 to have isolated an element number 72 from a sample of rare earth (elements 58–71) compounds. However, Niels Bohr believed that hafnium was more likely to be found along with zirconium than with the rare earths. D. Coster and G. von Hevesy, working in Bohr's laboratory in Copenhagen, showed in 1922 that element 72 was present in a sample of Norwegian zircon, an ore of zirconium. (The name hafnium comes from the Latin name for Copenhagen, Hafnia). (c) Solid zirconium dioxide, ZrO2, reacts with chlorine gas in the presence of carbon. The products of the reaction are ZrCl4 and two gases, CO2 and CO in the ratio 1:2. Write a balanced chemical equation for the reaction.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
568
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos