13. Liquids, Solids & Intermolecular Forces
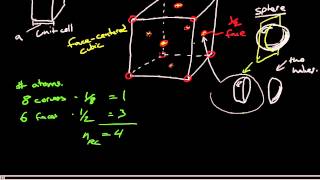
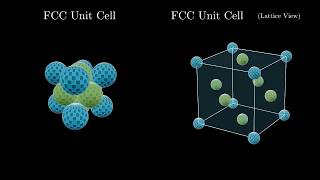
Face Centered Cubic Unit Cell
Problem 61b
Textbook Question
Textbook QuestionA particular form of cinnabar (HgS) adopts the zinc blende structure. The length of the unit cell edge is 5.852 Å. (b) The mineral tiemannite (HgSe) also forms a solid phase with the zinc blende structure. The length of the unit cell edge in this mineral is 6.085 Å. What accounts for the larger unit cell length in tiemmanite?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
517
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos