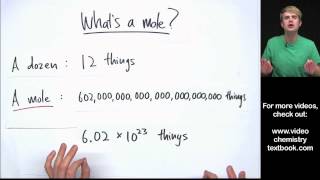2. Atoms & Elements
Mole Concept
Problem 89d
Textbook Question
Textbook QuestionCalculate the mass, in grams, of each sample. c. 1.8 * 1023 lead atoms
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
538
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos