2. Atoms & Elements
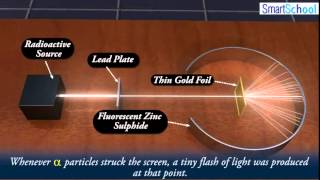
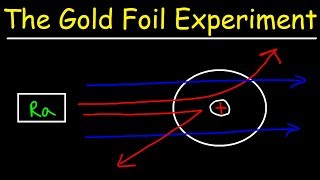
Rutherford Gold Foil Experiment

Get help from an AI Tutor
Ask a question to get started.
Problem 92
Textbook Question
Textbook QuestionPrior to Rutherford's gold foil experi-ment, the 'plum pudding' model of the atom represented atomic structure. In this model, the atom is composed of elec-trons interspersed within a positive cloud of charge. If this were the correct model of the atom, predict how the results of Rutherford's experiment would have been different. (a) The alpha particles would pass right through the gold foil with little to no deflection. (b) Most of the alpha particles would be deflected back toward the source. (c) Most of the alpha particles would be absorbed by the atom and neither pass through nor be deflected from the gold foil.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
355
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos