10. Periodic Properties of the Elements
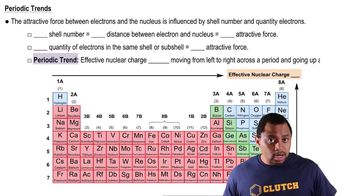
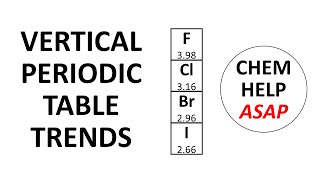
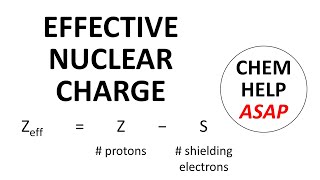
Periodic Trend: Effective Nuclear Charge

Get help from an AI Tutor
Ask a question to get started.
Problem 15
Textbook Question
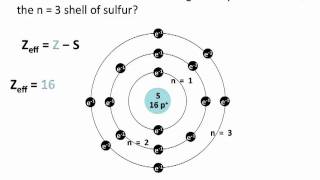
Textbook QuestionDetailed calculations show that the value of Zeff for the outermost electrons in Na and K atoms is 2.51+ and 3.49+, respectively. (e) Predict Zeff for the outermost electrons in the Rb atom based on the calculations for Na and K.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
787
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos