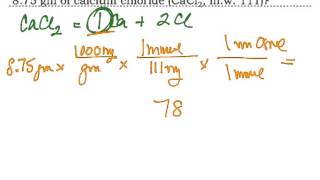6. Chemical Quantities & Aqueous Reactions
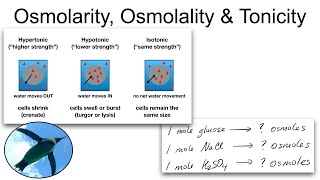
Osmolarity
Problem 123
Textbook Question
Textbook QuestionConsider two solutions, the first being 50.0 mL of 1.00 M CuSO4 and the second 50.0 mL of 2.00 M KOH. When the two solutions are mixed in a constant-pressure calorimeter, a precipitate forms and the temperature of the mixture rises from 21.5 to 27.7 °C. (a) Before mixing, how many grams of Cu are present in the solution of CuSO4?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
878
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos